RVLM (Recursive Vision-Language Model) is a unified framework for medical AI that replaces traditional single-pass inference with an iterative "generate-execute" REPL loop. By integrating RRouter for adaptive iteration depth, it achieves SOTA-level interpretability and efficiency on BraTS 2023 (Brain MRI) and MIMIC-CXR benchmarks without task-specific fine-tuning.
TL;DR
Medicine demands transparency, yet most AI models are "black boxes." RVLM (Recursive Vision-Language Model) changes the paradigm by replacing single-pass guesses with a verifiable Python-based reasoning loop. Coupled with RRouter, which scales the "thinking time" based on case complexity, this framework achieves human-like multi-step diagnostic reasoning that is fully auditable and efficient.
Context: Why "One-Shot" Isn't Enough for Radiology
When a radiologist looks at an MRI, they don't just glance and shout a diagnosis. They compare modalities (T1 vs. T2), zoom into suspicious regions, and measure volumes. Current VLMs (like GPT-4o or Gemini) try to do all this in a single "forward pass." This leads to two major issues:
- Interpretability Gap: You can't see why the model reached a conclusion.
- Resource Waste: The model spends the same "compute energy" on a clear-cut case as it does on a rare, complex tumor.
Methodology: The "Generate-Execute" Loop
RVLM embeds a multimodal model within a Visual REPL (Read-Eval-Print Loop). Instead of outputting text directly, the model writes Python code at each step to:
- Inspect: Specifically query different image modalities (e.g., looking for "dural tail signs" on T1ce).
- Manipulate: Crop regions of interest or compute "difference maps" between slices.
- Verify: Store observations in variables (
t1_desc,edema_check) to build a structured evidence chain.
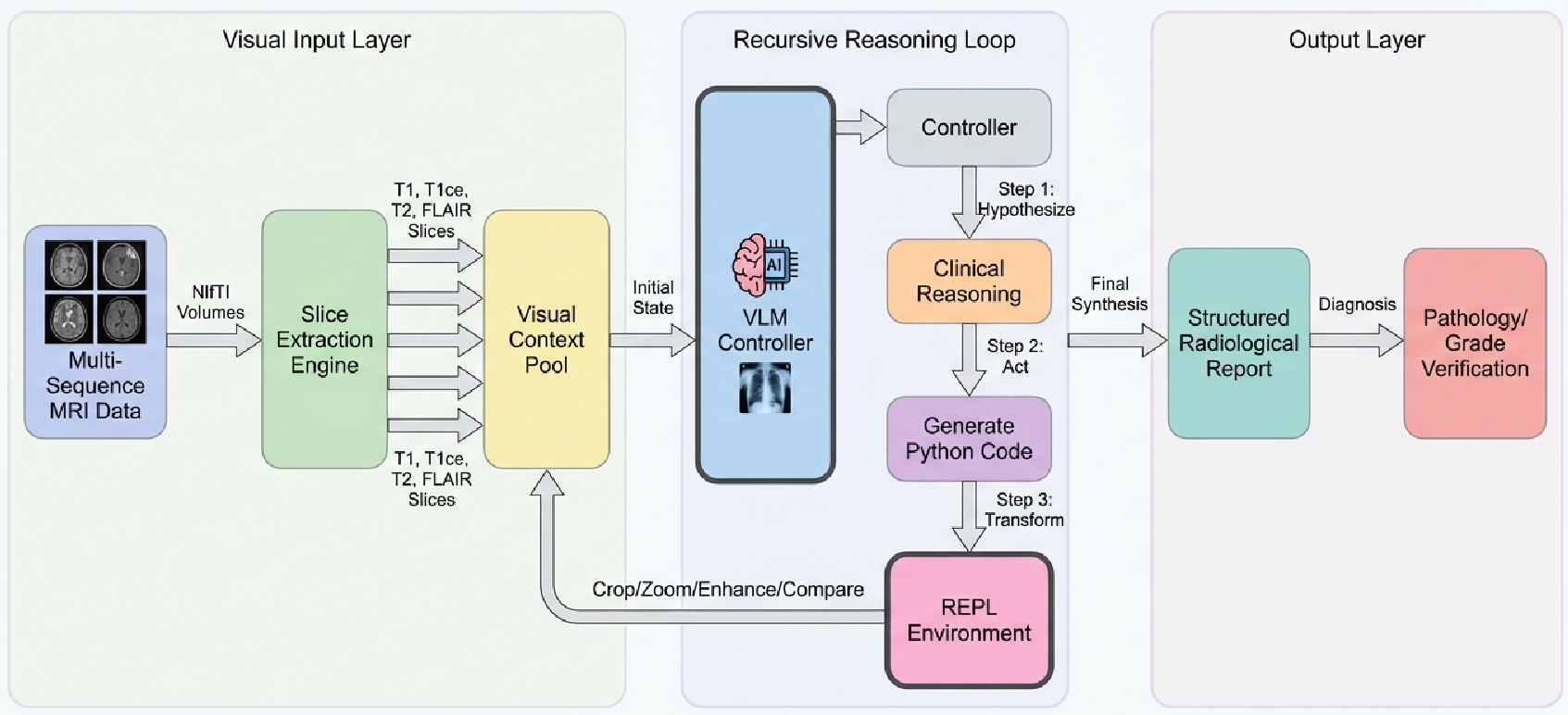 Figure 1: The RVLM System Architecture showing the iteration between the VLM controller and the persistent Python environment.
Figure 1: The RVLM System Architecture showing the iteration between the VLM controller and the persistent Python environment.
RRouter: Thinking Only as Much as Needed
To solve the "fixed-budget" problem, the authors introduced RRouter. It calculates a Complexity Score ($s$) based on:
- Label Entropy ($H$): How "messy" is the tumor's structure?
- Volume ($V$): How large is the lesion?
- Sub-region Count: How many different types of tissue are involved?
Simple cases get 3 iterations; complex ones get up to 6+. This mirrors clinical intuition: don't over-analyze a simple fracture, but take your time with a multifocal mass.
Experimental Highlights: BraTS & MIMIC-CXR
The framework was tested on Brain MRIs (BraTS) and Chest X-rays (MIMIC-CXR) using Gemini 2.5 Flash as the engine.
1. The Power of Cross-Modal Verification
In a notable case (BraTS-MEN-00008-000), RVLM detected a hyperintense ring on the FLAIR modality but noticed the segmentation mask showed 0.00cc of edema. It autonomously flagged this cross-modal discrepancy—a high-level reasoning task that single-pass models typically fail.
2. Efficiency Gains
RRouter successfully identified simple cases and capped the budget at 3 iterations, providing a 4x reduction in API costs and latency compared to a maximum-iteration ceiling, without losing diagnostic accuracy.
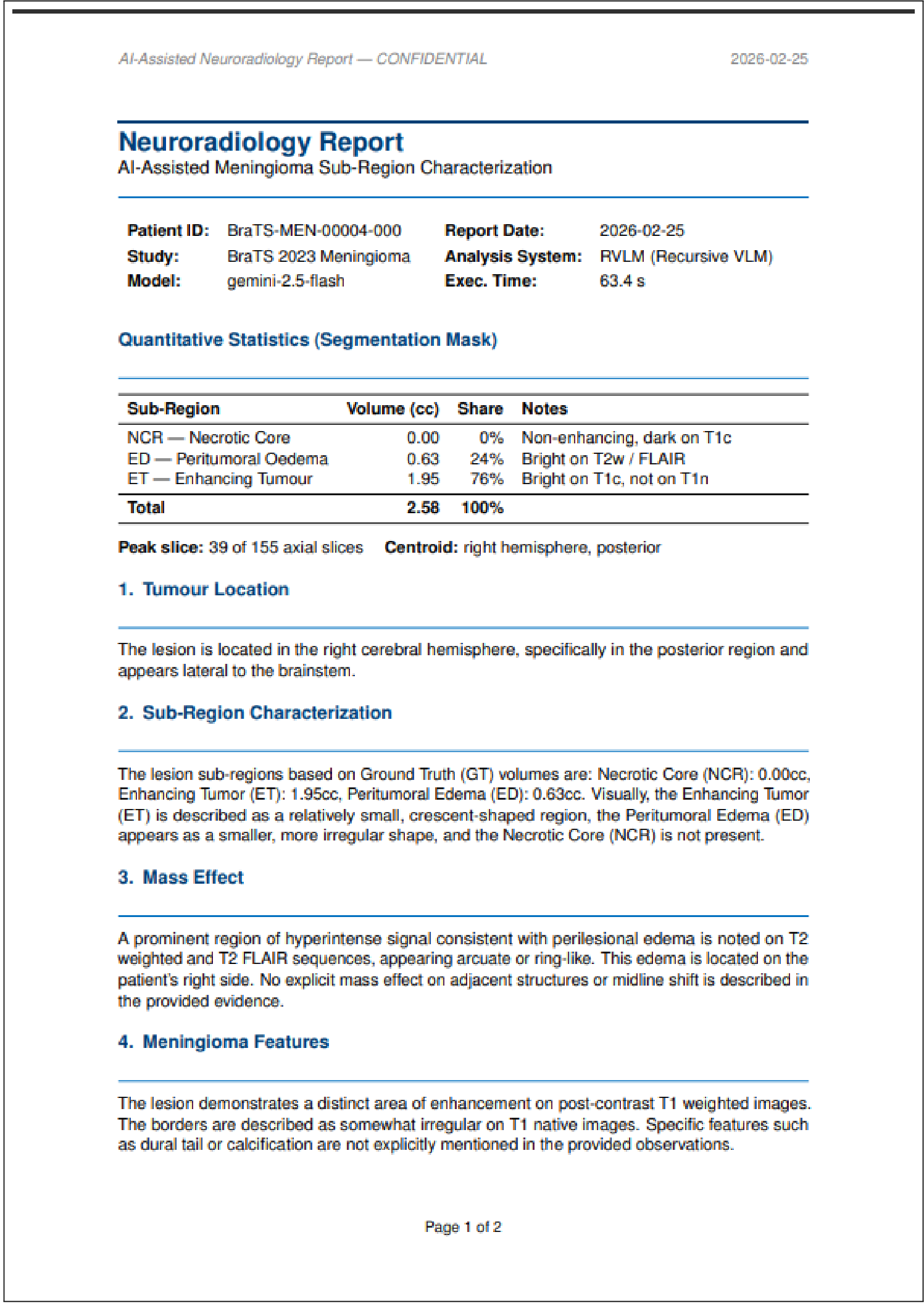 Figure 2: An auto-generated structured PDF report produced by RVLM, complete with an audit trail of the reasoning steps.
Figure 2: An auto-generated structured PDF report produced by RVLM, complete with an audit trail of the reasoning steps.
Critical Insight: Trust-by-Design
The most significant contribution of RVLM isn't just "higher accuracy"—it's Regulatory Readiness. By grounding every claim in executable code, RVLM provides:
- Traceability: You can re-run the exact code used to generate a diagnosis.
- Human Oversight: Clinicians can inspect the intermediate Python variables to see what the AI "saw" at iteration 2 vs. iteration 4.
- Compliance: It maps directly to the transparency requirements of the EU AI Act and ISO 42001.
Limitations & Future Path
While groundbreaking, the study is currently limited by a small qualitative sample size and 2D slice analysis. The authors point towards learned routers (using ML to predict complexity) and 3D volumetric reasoning as the next frontiers.
Conclusion
RVLM proves that we don't need to choose between performance and interpretability. By treating diagnostic reasoning as a recursive, adaptive process rather than a static prediction, we can build AI systems that clinicians can actually trust.
