This paper investigates the functional significance of Respiratory Sinus Arrhythmia (RSA) using a synchronization-dissipation model. By simulating the electrical and viscoelastic interactions in the cardiorespiratory system, the authors demonstrate that cardiac synchronization with respiration reduces energy dissipation in the pulmonary vasculature, potentially improving cardiac output by 10% to 55%.
Executive Summary
TL;DR
Why does our heart speed up when we inhale and slow down when we exhale? This phenomenon, known as Respiratory Sinus Arrhythmia (RSA), has puzzled physiologists for decades. This paper provides a compelling thermodynamic answer: Energy Efficiency. By modeling the heart and lungs as coupled dissipative oscillators, the researchers show that synchronization reduces mechanical power loss in the pulmonary vasculature by up to 10% in humans, directly easing the workload of the right ventricle.
Contextual Positioning
This work moves beyond the traditional view of RSA as a mere byproduct of autonomic reflexes. It positions the cardiorespiratory system within the framework of non-equilibrium physics, specifically "synchronization-dissipation" dynamics. It transitions RSA from a physiological curiosity to a vital SOTA (State-Of-The-Art) mechanism for hemodynamic optimization.
The Core Motivation: The Hidden Cost of Pumping
The right ventricle (RV) spends roughly 1/6th of its power on static vascular resistance. However, a staggering 40% of its power is lost to the oscillatory component of blood flow—the energy wasted as vessel walls stretch and contract.
The researchers identified a crucial Inductive Bias in the system:
- Lung Inflation causes axial strain (stretching) on pulmonary capillaries.
- Cardiac Systole (contraction) creates blood pressure surges that expand capillaries.
If these two rhythms are random, their mechanical strains add up destructively. But if they synchronize, the strains can compensate for each other, minimizing the total work the heart must perform.
Methodology: Modeling the "Biological Clock"
The authors utilized a dual-modeling approach to bridge the gap between electrical signaling and mechanical dissipation.
1. The Electrical Pacer (Hodgkin-Huxley Model)
To replicate the brainstem's control, they used the Hodgkin-Huxley equations to drive a neuronal oscillator. This oscillator's frequency is modulated by "injected current" representing lung stretch receptors.
- Inspiration: High current $\rightarrow$ Higher heart rate.
- Expiration: Low current $\rightarrow$ Lower heart rate.
2. The Mechanical Load (Viscoelastic Dissipation)
The power dissipation $p_v$ in the capillary wall is defined by the viscosity $\eta$ and the variance of the strain rate $\dot{\epsilon}$: $$\langle p_v \rangle = \eta {Var(\dot{\epsilon}_R) + Var(|\dot{\epsilon}_H|) - Cov(\dot{\epsilon}_R, |\dot{\epsilon}_H|)}$$ The key insight here is the Covariance term. If the rhythms are synchronized, the covariance is positive, and the total power dissipation drops.
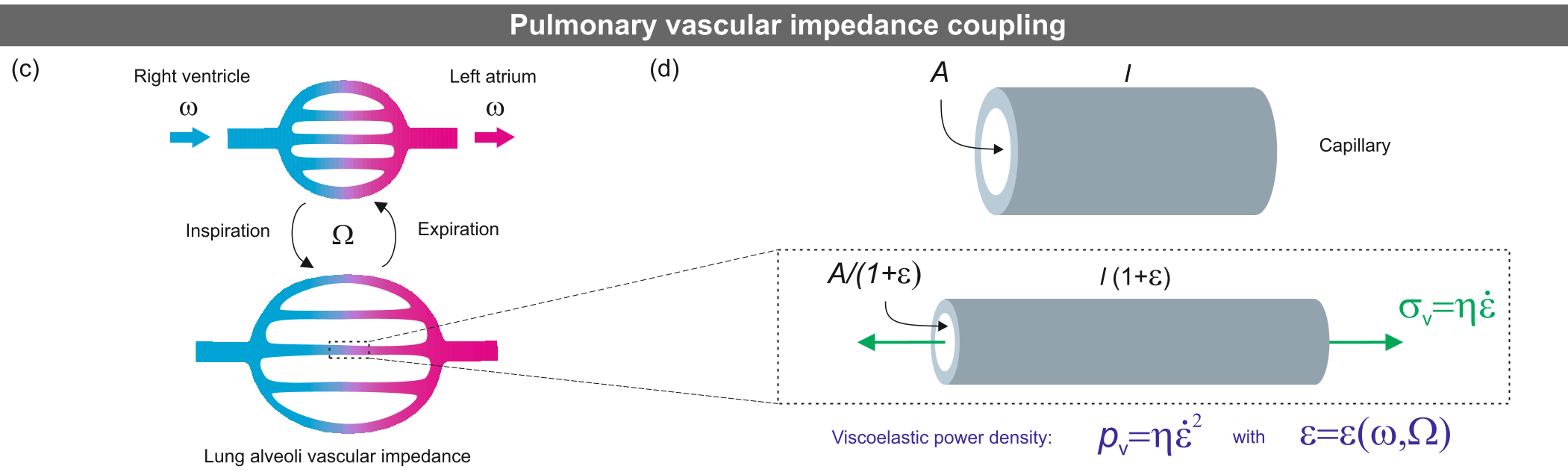 Fig 1: Schematic showing (a) CNS coupling, (b) the neuronal pacemaker, and (c) the viscoelastic interaction at the alveoli.
Fig 1: Schematic showing (a) CNS coupling, (b) the neuronal pacemaker, and (c) the viscoelastic interaction at the alveoli.
Critical Results: Entering the Arnold Tongues
The simulation revealed that synchronization doesn't happen at every frequency. Instead, it occurs in specific regions called Arnold Tongues—frequency bands where the heart-to-breath ratio is a stable integer (e.g., 3:1 or 4:1).
The Power Savings Map
The researchers generated a "Power Map" overlaying synchronization regions with energy gains:
- Human Physiological State: At a 3:1 ratio (typical for rest), the model predicts a 10% reduction in power loss.
- Max Efficiency: In high-coupling scenarios (1:1 ratio), energy savings can reach 55%.
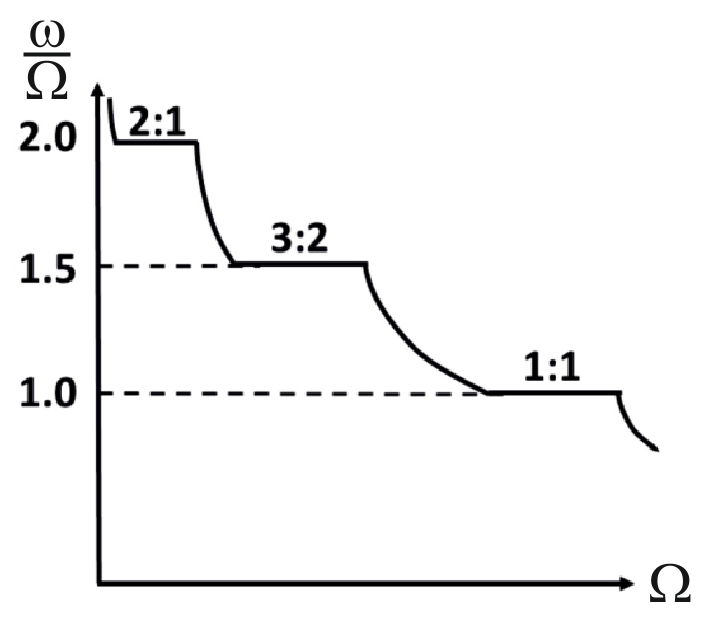 Fig 2: The Arnold Tongues (frequency locking) perfectly coincide with the dark blue areas of minimum power dissipation.
Fig 2: The Arnold Tongues (frequency locking) perfectly coincide with the dark blue areas of minimum power dissipation.
Comparison with In-Vivo Data
The theoretical 10% efficiency gain is consistent with experimental evidence using "bionic" pacemakers. In sheep and pig models of heart failure, restoring RSA increased cardiac output by 17-20%. The discrepancy suggests RSA also benefits gas exchange and systemic circulation in ways not fully captured by the mechanical model alone.
Deep Insights & Future Outlook
Why This Matters for Technology
Current pacemakers are "monotonic"—they pulse at a fixed, robotic rate. This study provides the physics-based justification for adaptive, nonlinear pacemakers. By sensing the respiratory rhythm and re-synchronizing the heart beat, we can treat heart failure not just by "pacing" the heart, but by making its environment more energy-efficient.
Limitations
- Geometric Simplification: The model assumes uniform capillary behavior, whereas the lung is a complex, hierarchical network.
- Aperiodicity: While the authors address aperiodic breathing, real-world heart-rate variability is even more chaotic than the current model.
Conclusion
This paper elegantly demonstrates that RSA is not a "noise" in the system but a sophisticated optimization strategy. By exploiting synchronization-dissipation, the body reduces the "dynamic stress" on the heart, proving once again that biological evolution is the ultimate engineer.
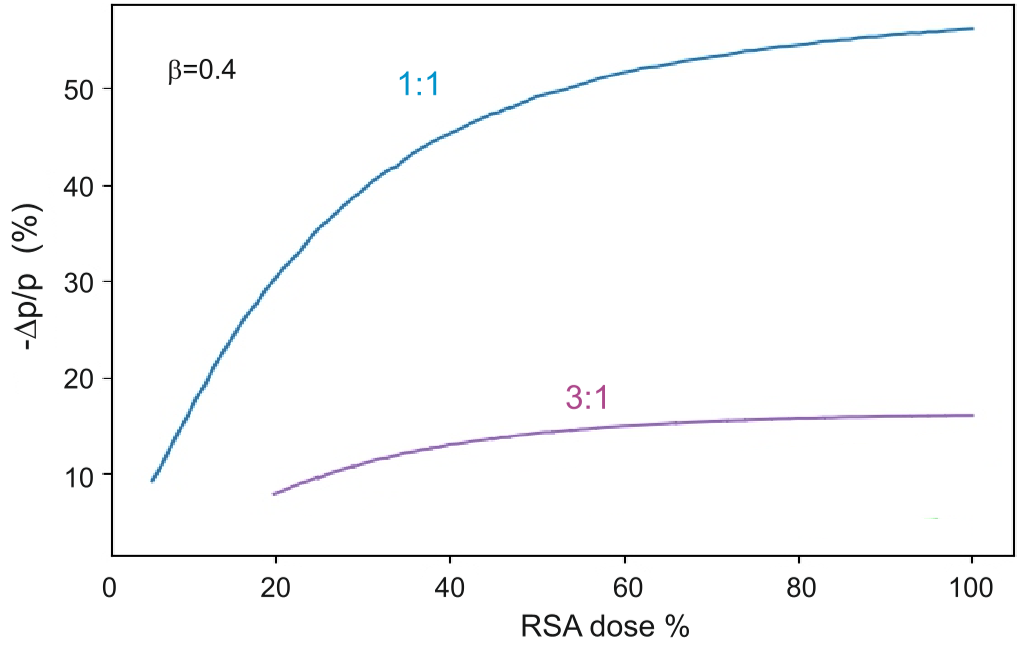 Fig 3: Power gain increases proportionally with the "RSA dose," highlighting the therapeutic potential of heart rate variability.
Fig 3: Power gain increases proportionally with the "RSA dose," highlighting the therapeutic potential of heart rate variability.
